Documentation Index
Fetch the complete documentation index at: https://reactionrepo.mintlify.app/llms.txt
Use this file to discover all available pages before exploring further.
Goals
hiTypes of Arrows in Mechanisms
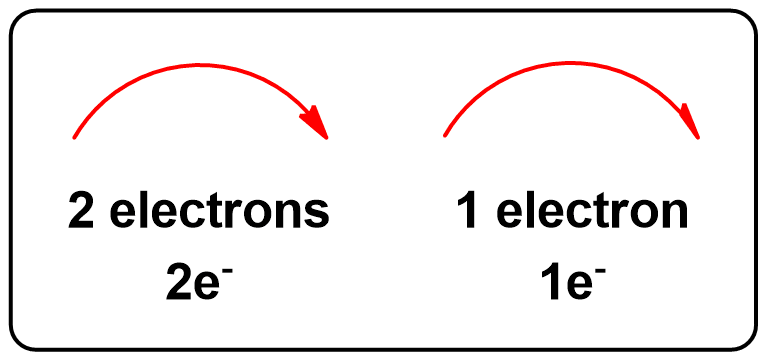
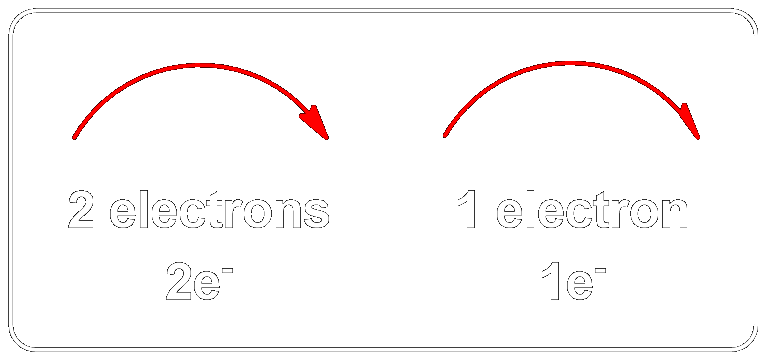
The Two Types of Arrows used in Mechanisms.
Representation of electron movement: Representation of electron movement: two-electron (1 bond) processes (Full-arrow →) vs. one-electron (half bond) processes (Half-arrow ↷).
Full Arrows (→)
A full curved arrow represents the movement of two electrons, usually in bond formation or breaking. This applies to:- Nucleophilic attacks
- Resonance structures
- Electrophilic and nucleophilic reactions
- Nucleophiles attack electrophiles by donating an electron pair to form a new bond.
- Electrophiles receive the nucleophillic attack to accept an electron pair to form a new bond.
Nucleophiles donate electron pairs (electron-rich) vs. Electrophiles accept electron pairs (electron-poor).
Half Arrows (↷)
A half-arrow represents the movement of one electron, commonly seen in:- Radical reactions
- Homolytic bond cleavage
- Radical propagation steps
There will be lots of examples in the entries of Reaction Repo of all reactions mentioned
Applications of the Arrows
Bond Formation
Full Arrow
Half Arrow
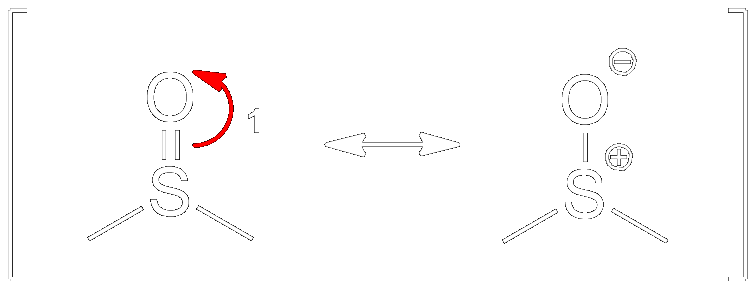
Forming Resonance Structures
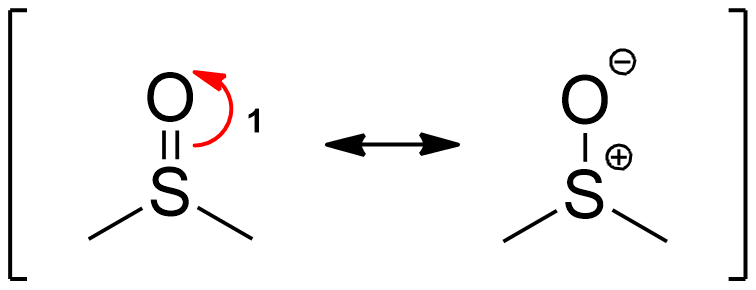
Resonance forms of DMSO.
DMSO is capable of undergoing resonance. The first step (1) towards the Swern Oxidation